PRECISION REGENERATIVE MEDICINE
Regenerative medicine offers significant promise to restore tissue functions from damage and to improve quality of life. Tissue damage often results in fibrosis and impaired organ function, which accounts for up to 45% of all deaths in the developed world. Despite an exponential growth in various approaches to regenerate tissues, clinical adoption has been limited due to incomplete mechanistic understanding and lack of control in cell functions. We are interested in understanding how cells build and remodel their environments in physiology and disease. Our team is pursuing multidisciplinary approaches to understand the fundamentals in physical biology of regenerative processes and to leverage these insights by precisely directing donor cells and modulating the host response to treat tissue injury, degeneration and fibrosis. Target organs include the bone marrow, bones, vasculature, and lungs.
Regenerative medicine offers significant promise to restore tissue functions from damage and to improve quality of life. Tissue damage often results in fibrosis and impaired organ function, which accounts for up to 45% of all deaths in the developed world. Despite an exponential growth in various approaches to regenerate tissues, clinical adoption has been limited due to incomplete mechanistic understanding and lack of control in cell functions. We are interested in understanding how cells build and remodel their environments in physiology and disease. Our team is pursuing multidisciplinary approaches to understand the fundamentals in physical biology of regenerative processes and to leverage these insights by precisely directing donor cells and modulating the host response to treat tissue injury, degeneration and fibrosis. Target organs include the bone marrow, bones, vasculature, and lungs.
Research areas
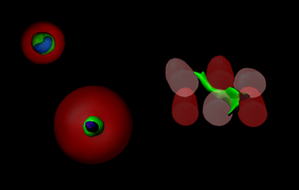
Precision niche design
We are combining biomaterial design with microtechnologies to build instructive niches that can precisely guide cells to regenerate tissues.
Cho et al. Deterministic single cell encapsulation in asymmetric microenvironments to direct cell polarity. Adv. Sci. 2023; 10: 2206014.
Wong et al. Controlled deposition of 3D matrices to direct single cell functions. Adv. Sci. 2020; 7: 2001066.
Devine, Vijayakumar, Wong et al. Hydrogel micropost arrays with single post tunability to study cell volume and mechanotransduciton. Adv. Bio. 2020; 4: 2000012.
We are combining biomaterial design with microtechnologies to build instructive niches that can precisely guide cells to regenerate tissues.
Cho et al. Deterministic single cell encapsulation in asymmetric microenvironments to direct cell polarity. Adv. Sci. 2023; 10: 2206014.
Wong et al. Controlled deposition of 3D matrices to direct single cell functions. Adv. Sci. 2020; 7: 2001066.
Devine, Vijayakumar, Wong et al. Hydrogel micropost arrays with single post tunability to study cell volume and mechanotransduciton. Adv. Bio. 2020; 4: 2000012.

Engineering host remodeling
We are developing instructive materials to control how cells remodel host tissues after damage.
Wong et al. Inhibition of aberrant tissue remodeling by mesenchymal stromal cells singly coated with soft gels presenting defined chemomechanical cues. Nat. Biomed. Eng. 2022; 6: 54-66.
Wong et al. Soft extracellular matrix enhances inflammatory activation of mesenchymal stromal cells to induce monocyte production and trafficking. Sci. Adv. 2020; 6: eaaw0158.
We are developing instructive materials to control how cells remodel host tissues after damage.
Wong et al. Inhibition of aberrant tissue remodeling by mesenchymal stromal cells singly coated with soft gels presenting defined chemomechanical cues. Nat. Biomed. Eng. 2022; 6: 54-66.
Wong et al. Soft extracellular matrix enhances inflammatory activation of mesenchymal stromal cells to induce monocyte production and trafficking. Sci. Adv. 2020; 6: eaaw0158.
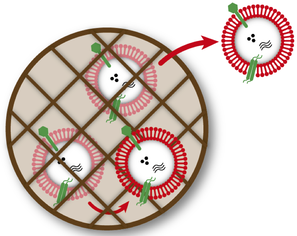
Extracellular nanoscale mediators in context
We are leveraging engineered biomaterials to investigate how cell-secreted extracellular nanoscale mediators with regenerative functions are secreted from cells and transported in tissues.
Debnath, Las Heras et al. Extracellular vesicle-matrix interactions. Nat. Rev. Mater. 2023.
Lenzini et al. Cell-matrix interactions regulate functional extracellular vesicle secretion from mesenchymal stromal cells. ACS Nano. 2021; 15: 17439-17452.
Lenzini et al. Matrix mechanics and water permeation regulate extracellular vesicle transport. Nat. Nano. 2020; 15: 217-223.
We are leveraging engineered biomaterials to investigate how cell-secreted extracellular nanoscale mediators with regenerative functions are secreted from cells and transported in tissues.
Debnath, Las Heras et al. Extracellular vesicle-matrix interactions. Nat. Rev. Mater. 2023.
Lenzini et al. Cell-matrix interactions regulate functional extracellular vesicle secretion from mesenchymal stromal cells. ACS Nano. 2021; 15: 17439-17452.
Lenzini et al. Matrix mechanics and water permeation regulate extracellular vesicle transport. Nat. Nano. 2020; 15: 217-223.
Institution
Funding sources



